Nitrogen Efficiency
It is hard to escape from the fact that the cost of nitrogen fertilisers has increased significantly due to a range of factors placing strain on both supply and demand globally. With this in mind, it is useful to reconsider why we use nitrogen and how to use nitrogen efficiently to reduce costs and environmental harm and improve plant growth.
Why are prices for nitrogen fertilisers increasing?
The price of nitrogen fertilisers is significantly influenced by costs of natural gas due to the importance of this resource in the fertiliser production process.
Globally, the majority of synthetic fertilisers are produced industrially using the Haber-Bosch process in which nitrogen (N2) from the air is combined with hydrogen (H2) from natural gas to produce liquid ammonia (NH3). This energy intensive process utilises fossil fuels to achieve the high temperatures of 400-500°C and pressures of 150-300 bar that are required. The ammonia is then used to make nitric acid which can be combined with ammonia to create ammonium nitrate or with liquid carbon dioxide to create urea.
In total it is estimated that production of nitrogen fertilisers accounts for three to five percent of the global annual natural gas consumption (European Commission, 2019). The reliance on energy intensive processes and fossil fuels, in particular natural gas, is the primary driver for nitrogen fertiliser prices.
Why is nitrogen important for plant growth?
Nitrogen is a key component of proteins which help plants to grow by providing structural support for cells, helping cells to interact with their environment, and reducing the energy requirements for biochemical reactions. Proteins also transport nitrogen through the plant and are nitrogen donors for other fundamental compounds such as:
• Nucleic acids: The building blocks of DNA and RNA
• Hormones: Chemicals which control activities within the plant
• Chlorophyll: The pigments that enable plants to create food through photosynthesis
Proteins are formed from the bonding of amino acids. Each amino acid has a basic structure with a central carbon atom which is bonded to an amino group (NH2), a carboxyl (acid) group (COOH), and a hydrogen atom. The final component is an R group which determines the unique properties of each of the 20 different amino acids.
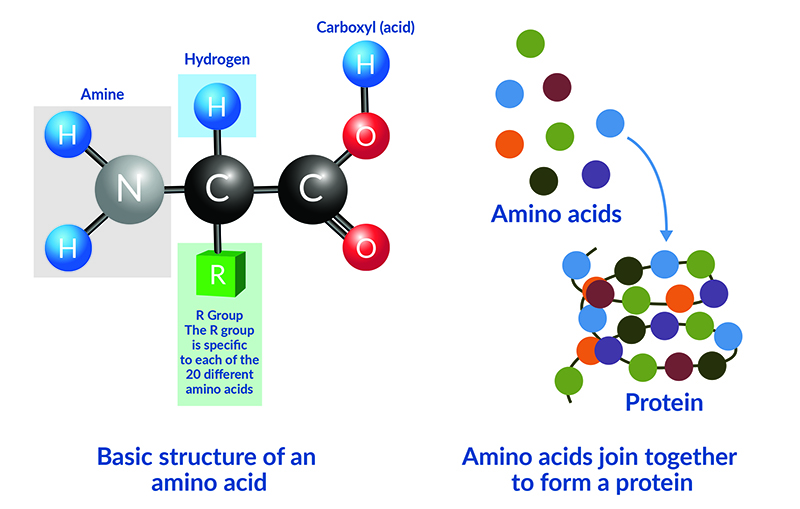
The nitrogen that is used as a component of amino acids and proteins is generally taken up from the soil in the form of either nitrate (NO3-) or ammonium (NH4+).
Nitrates are the preferred form and can either be stored in the root for later use or translocated to the shoot where it is assimilated into amino acids. When ammonium is taken up from the soil assimilation generally occurs in the plant root following uptake (Tischner, 2000).
Why is excess nitrogen harmful?
Applying excess nitrogen is not only a waste of available resources and money but can also lead to problems with the plants it is intended to support, to the wider environment and to human health.
Plants
Excessive levels of nitrogen fertiliser can give foliage a weak physical structure which is susceptible to both abiotic stresses such as drought, heat or cold and to biotic stresses such as disease and insect damage. Whilst there is no specific nitrogen toxicity effect, the physical symptoms described above can cause significant damage if excess nitrogen conditions are allowed to continue. In addition, it is known that ammonium can induce toxicity in plants when conditions limit the ability of the growing media to convert ammonium to nitrate (Handreck and Black, 2002). Conditions that can induce this are related to low soil oxygen levels (i.e. compaction, drainage etc) (Gelernter et al., 2001) and to reduced photosynthetic potential (dull, cool weather) (Handreck and Black, 2002). Plants can become wilted and stunted (Handreck and Black, 2002).
Soils
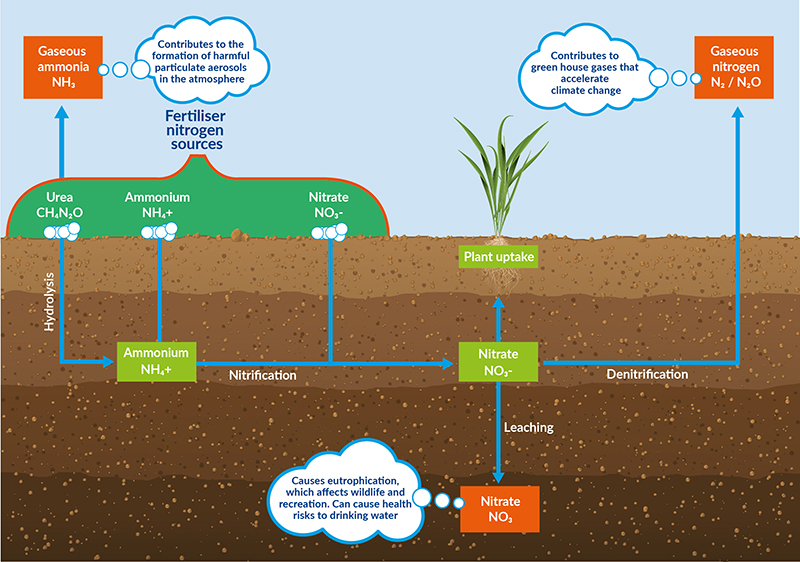
Excess applications of ammonium can lead to acidification of soils from the generation of hydrogen ions (H+) during conversion to nitrate. If the nitrogen is taken up by plants, hydroxide ions (OH ) combine with the hydrogen ions to form water (H2O). Acidification is exacerbated when the nitrogen is not taken up by plants, and no hydroxide ions are released to combine with the hydrogen, creating a net increase in hydrogen ions leading to an increase in acidity.
Water
Nitrogen that is not taken up by plants can leach from soils into water sources, affecting drinking water quality and creating pollution issues such as eutrophication.
Drinking water quality is compromised when nitrogen leaches into water sources because excessive nitrate can restrict oxygen transport in the bloodstream. Nitrate vulnerable zones (NVZ) were devised as a method of reducing nitrates in drinking water by requiring landowners to follow certain rules which minimise nitrate losses to water. NVZ designations are in force over some areas in the east of Scotland and the east of Wales. A large proportion of England is designated as a NVZ (UKSO, 2022).
Eutrophication can be caused when excess nitrogen leaches into water leading to excessive plant growth. This results in detrimental changes to the quality of the water and its value as a habitat.
Air
Release of gaseous ammonia (NH3) following applications of urea has adverse effects on human health as gaseous ammonia reacts with other chemicals in the air to form particulate matter.
Nitrates that are not taken up by the plant can undergo a process called de-nitrification which results in the formation of gaseous forms of nitrogen such as nitrous oxide N2O. Gaseous nitrous oxide (N2O) is a potent greenhouse gas which contributes to climate change.
How can nitrogen be used efficiently?
Due to the importance of nitrogen to plant health and growth, it's potential for harm to the environment and to public health, alongside price volatility it is increasingly important to minimise nitrogen losses whilst maximising the value from nitrogen inputs. There are a range of strategies that can be utilised to achieve this.
Nitrogen sources
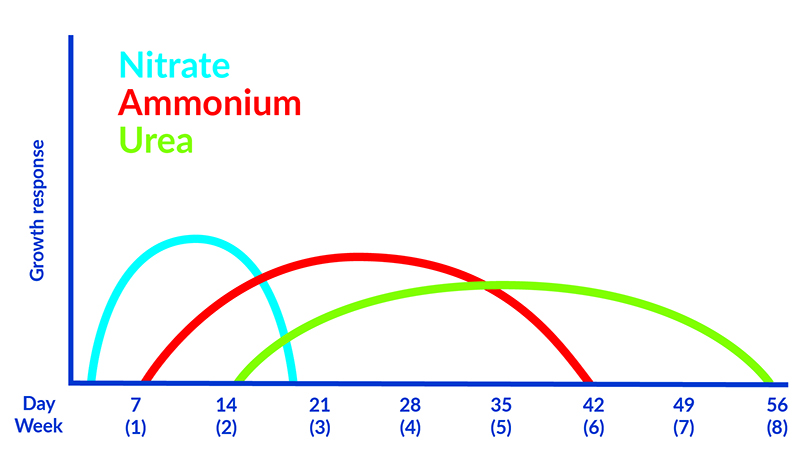
Different nitrogen sources are converted to plant available forms over different timescales. Nitrate followed by ammonium are the most plant available forms of the nutrient. When other sources of nitrogen are supplied, for example urea, it must be converted to these plant available forms before it becomes useful to the plant.
The aim is to match nitrogen sources with plant requirements over time. For example, a nitrate fertiliser may provide an initial hit of nitrogen that is rapidly depleted whereas urea will take a period of time to be converted into a useable form but may be available for plant uptake for longer. Many fertilisers combine nitrogen sources to provide a consistent amount of nitrogen over the expected release period.
Fertiliser formulations
In addition to containing different nitrogen sources, fertilisers are available in different formulations which affect the speed and longevity of nitrogen delivery to the plant. This in turn affects the likelihood of losses to the environment.
Liquid fertilisers
Liquid fertilisers generally release nutrients rapidly with typical longevity around 2-4 weeks. They excel at providing fine nutrient control determined by plant requirements.
Granular fertilisers
Conventional granular fertilisers have a typical nitrogen release period of 2-6 weeks. Organo-mineral, slow-release, or controlled release use different methods to reduce losses to the environment with typical nitrogen release periods of 6-12 weeks, 6-12 weeks, and 12+ weeks respectively:
• Organo-mineral fertilisers: gradually convert nitrogen from an organic form to a plant available form such as nitrate or ammonium.
• Slow-release fertilisers: gradually convert chemically bound urea to a plant available nitrogen form. Nitrification/urease inhibitors might also be used.
• Controlled release fertilisers: utilise coatings to determine release rate by water penetration and temperature.
Cultural management strategies
Fertiliser application
Soil nitrate and ammonium levels are extremely variable over short periods of time and therefore they are not usually tested for alongside other nutrients in soil analyses. Even during the time period that it takes for a soil sample to get from the soil to the lab and tested there is likely to be a considerable change in the amount of nitrogen present. Because of this, nitrogen application recommendations are usually based on other known factors such as grass species, turf use and soil type.
Nitrogen fertilisers should only be used when plants are growing actively. The Code of Good Agricultural Practices for Farmers, Growers and Land Managers states that nitrogen fertilisers should not be applied to grass between 15 September and 15 January unless there is a specific crop requirement at this time (DEFRA, 2009). This helps to ensure that nitrogen fertilisers are used during active plant growth to coincide with plant nitrogen requirements.
Growth driven methods provide further optimisation to ensure that nutrient applications are made prior to observable nutrient deficiencies and do not exceed plant requirements. Growth driven methods measure plant nitrogen use to direct management inputs. Suitable methods include measuring clipping volumes to approximate nutrient removal (Kreuser, 2018), or directly measuring leaf nitrogen levels (Ericsson, et al., 2021). Fertilisation inputs can then be increased/decreased in response to sustained deviations from measured levels.
3.1.1 Mowing
Nitrate uptake can be impaired following mowing and therefore avoiding nutrient applications immediately after mowing could improve uptake (Bowman, 1987).
3.1.2 Irrigation
Opportunities for leaching can be reduced if irrigation rates and frequencies do not allow water to reach beyond the active rooting depth (Barton & Colmer, 2006).
3.1.3 Use of complementary products
Complementary products are those which facilitate better uptake or use of nutrients in the plant:
• Humates can contribute to improved nitrogen use efficiency by stimulating the H+ -ATPase enzyme which acts as a proton pump to increase movement of nutrients from the soil solution into the xylem.
• Amino acids utilise nitrogen as an essential component, but the synthesis of amino acids is a very energy intensive process. Therefore, providing a supply of amino acids to the plant helps to reduce energy use on this process, resulting in an increase in energy for other processes such as growth.
• Carbon sources can increase or maintain the carbon to nitrogen ratio in the soil, ensuring there is an energy source to fuel the microorganisms that convert nitrogen sources into plant available compounds.
• Plant growth regulators can reduce plant nitrogen requirements simply because the amount of growth is reduced. If nitrogen application is measured to match nitrogen removal through mowing, a reduction in mowing requirements will result in a reduction in the nitrogen required (Soldat & Kreuser, 2012).
Dr Abigail Graceson - Technical Manager, Agrovista Amenity